Skrypt forum został zaktualizowany. W przypadku problemów z logowaniem prosimy o skorzystanie z opcji Nie pamiętam hasła.
Wybarwianie roślin
Moderator: Administracja
Re: Wybarwianie roślin
Darek, szukaj pod tagami "photosynthesis C4 sodium". Nie znajdę teraz linka z którego akurat korzystałem ale wydawało mi się, że go wklejałem. Będę go szukał jeszcze. Ale nawet z poniższego linku najważniejsze jest zdanie.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC366054/
"It is concluded from these results and previously reported work that sodium is generally essential for species having the C4 pathway but not for species with the C3 pathway."
Czy teraz rozumiesz czemu uważam, że wszystkie rośliny przeprowadzają fotosyntezę C4 i wymagają sodu jako mikroskładnika? No właśnie ja w kranie też mam sód i problem bąblowania kiedyś u mnie nie istniał.
Ale nie wpadłem na pomysł by przy mineralizacji RO dodawać sód. Dopiero analiza składu podłoża i działania podłoży jono uzmysłowiła mi potrzebę jego dozowania. Ale nie tyle co potasu. Tu mnie wpuścili w maliny.
W jakimś powerponcie przy szukaniu "fotosynteza C4" znalazłem info, że zwykła egeria jest C4. U mnie tylko jakiś wątrobowiec był C3 i tylko on bąblował.
Typoszereg siły wiązań już wklejałem. Faktycznie sód jest słabo związany. Ale u nas w akwa na szczęście nie ma zjawiska wymywania. Zasilania pierwiastkami z wód gruntowych poprzez kapilarność gruntu też nie ma. Nie wiem jak u Was w mieście ale u mnie po spadówie liści zgarniają je i wywożą. Dla mnie to karygodne bo najlepszym rozwiązaniem jest 5% roztwór mocznika i bakterie rozłożą liście błyskawicznie. I cykl pierwiastków niemobilnych byłby zachowany. Ale skądś na wiosnę liście drzew się zazieleniają więc dostawa pierwiastków jakaś się odbywa.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC366054/
"It is concluded from these results and previously reported work that sodium is generally essential for species having the C4 pathway but not for species with the C3 pathway."
Czy teraz rozumiesz czemu uważam, że wszystkie rośliny przeprowadzają fotosyntezę C4 i wymagają sodu jako mikroskładnika? No właśnie ja w kranie też mam sód i problem bąblowania kiedyś u mnie nie istniał.
Ale nie wpadłem na pomysł by przy mineralizacji RO dodawać sód. Dopiero analiza składu podłoża i działania podłoży jono uzmysłowiła mi potrzebę jego dozowania. Ale nie tyle co potasu. Tu mnie wpuścili w maliny.
W jakimś powerponcie przy szukaniu "fotosynteza C4" znalazłem info, że zwykła egeria jest C4. U mnie tylko jakiś wątrobowiec był C3 i tylko on bąblował.
Typoszereg siły wiązań już wklejałem. Faktycznie sód jest słabo związany. Ale u nas w akwa na szczęście nie ma zjawiska wymywania. Zasilania pierwiastkami z wód gruntowych poprzez kapilarność gruntu też nie ma. Nie wiem jak u Was w mieście ale u mnie po spadówie liści zgarniają je i wywożą. Dla mnie to karygodne bo najlepszym rozwiązaniem jest 5% roztwór mocznika i bakterie rozłożą liście błyskawicznie. I cykl pierwiastków niemobilnych byłby zachowany. Ale skądś na wiosnę liście drzew się zazieleniają więc dostawa pierwiastków jakaś się odbywa.
Pozdrawiam
Michał
Michał
-
cob_ra
- Posty: 287
- Rejestracja: 01 lut 2014, 9:45
- Imię: Marek
- Lokalizacja: Skierniewice
- Akwarium: Roślinniak 450l
- Płeć:
- Wiek: 40
Re: Wybarwianie roślin
Nie no, kurczę, ja nie mam siły, czasu i zaparcia, aby tak wnikać w każdy składnik i powiązania. Dla mnie to ma po prostu działać.
Fajnie, że tak mocno wnikacie w temat.
Fajnie, że tak mocno wnikacie w temat.
624l
- duch amazonski
- Posty: 1932
- Rejestracja: 03 wrz 2014, 19:44
- Imię: Darek
- Lokalizacja: LUBLIN
- Płeć:
- Wiek: 49
Re: Wybarwianie roślin
Michale, ostatnio znalazłem: https://academic.oup.com/jxb/article/65/3/849/524705
Wg. tego co czytam w różnych artykułach to Na należy dawkować raczej jako mikro, uniknąć wpadania do akwarium się się nie da. "Większe" ilości sodu szkodzą. Niektóre cryptocoryny przy dużej ilości Ca wstrzymują wzrost, myślę, że sód powoduje podobne "efekty". Oczywiście są rośliny z grupy c4 które tego sodu potrzebują więcej niż mikro dawki, ale to kilka % ogółu.
Wg. tego co czytam w różnych artykułach to Na należy dawkować raczej jako mikro, uniknąć wpadania do akwarium się się nie da. "Większe" ilości sodu szkodzą. Niektóre cryptocoryny przy dużej ilości Ca wstrzymują wzrost, myślę, że sód powoduje podobne "efekty". Oczywiście są rośliny z grupy c4 które tego sodu potrzebują więcej niż mikro dawki, ale to kilka % ogółu.
Prawdziwy mężczyzna płacze tylko wtedy, gdy szampon szczypie w oczka.
Re: Wybarwianie roślin
Ależ ja się z Tobą zgadzam. To mikroelement. Ale powinien być. Źle z interpretowałem podłoże bo tam, moim zdaniem rzecz jasna, sód służy do blokowania potasu, bo podłoże wychwyci dwukationowe i lecisz na oparach Ca i Mg.
Da się uniknąć. Nigdy nie dawałem sodu z żadnej soli. Na wysokim potasie dawałem bardzo mało mikro bo więcej nie potrzeba było i z nawozu mikroelementowego chelatowanego sodu było mało. Mikroelementy z soli w ogóle sodu nie miały.
Nie wiem jaki poziom jest bezpieczny dla roślin. Z pewnością antagonistami sodu jest K, Mg, Ca. Zatem będę musiał szukać proporcji by ciut tego sodu wpadło do rośliny. Bo to, ze miałem potas pomiędzy Ca a Mg nie oznacza, że był w roślinie w odpowiedniej ilości.
I jako ciekawostkę, być może znów źle interpretuje, ale spadek NO3 do zera to był całkowity zanik fotosyntezy. Czyli roślina transportuje węgiel w postaci asparaginianu a nie jabłczanu bo ten drugi nie ma azotu w swojej budowie. Tak se kombinuje.
Da się uniknąć. Nigdy nie dawałem sodu z żadnej soli. Na wysokim potasie dawałem bardzo mało mikro bo więcej nie potrzeba było i z nawozu mikroelementowego chelatowanego sodu było mało. Mikroelementy z soli w ogóle sodu nie miały.
Nie wiem jaki poziom jest bezpieczny dla roślin. Z pewnością antagonistami sodu jest K, Mg, Ca. Zatem będę musiał szukać proporcji by ciut tego sodu wpadło do rośliny. Bo to, ze miałem potas pomiędzy Ca a Mg nie oznacza, że był w roślinie w odpowiedniej ilości.
I jako ciekawostkę, być może znów źle interpretuje, ale spadek NO3 do zera to był całkowity zanik fotosyntezy. Czyli roślina transportuje węgiel w postaci asparaginianu a nie jabłczanu bo ten drugi nie ma azotu w swojej budowie. Tak se kombinuje.
Pozdrawiam
Michał
Michał
Re: Wybarwianie roślin
Miałem jakiś czas temu Ca- 35 K-30 Mg--3 przy teoretycznie zerowych fosforanach Obniżyłem K do 20 i zaczeło coś bąblować czyli K ma wpływ na Na co by potwierdzało że mamy rośliny C4
Fosfor ma praktycznie wpływ na całe mikro podnosząc fosfor tez przestaje bąblować czyli tez może wpływać na Na
Jezeli mamy niski Ca niski Mg to pozostaje K i P
Ps
Znacie jakiś gotowy nawóz mikro bez żelaza ?
Fosfor ma praktycznie wpływ na całe mikro podnosząc fosfor tez przestaje bąblować czyli tez może wpływać na Na
Jezeli mamy niski Ca niski Mg to pozostaje K i P
Ps
Znacie jakiś gotowy nawóz mikro bez żelaza ?
Re: Wybarwianie roślin
URL=upload/u4977_1636033092.jpg] [/URL]
[/URL]
Temat wybarwienia roślin więc mam pytanie czy ten pełzacz te cienkie igiełki to ma od CO2 ? Za plecami ma dyfuzor
 [/URL]
[/URL]Temat wybarwienia roślin więc mam pytanie czy ten pełzacz te cienkie igiełki to ma od CO2 ? Za plecami ma dyfuzor
- duch amazonski
- Posty: 1932
- Rejestracja: 03 wrz 2014, 19:44
- Imię: Darek
- Lokalizacja: LUBLIN
- Płeć:
- Wiek: 49
Re: Wybarwianie roślin
Wpływ pH i zasolenia na fotosyntezę u moczarki kanadyjskiej.
https://biologhelp.pl/wplyw-ph-zasoleni ... ologicznej
https://biologhelp.pl/wplyw-ph-zasoleni ... ologicznej
Prawdziwy mężczyzna płacze tylko wtedy, gdy szampon szczypie w oczka.
Re: Wybarwianie roślin
Już teraz wiem skąd się wzięło powiedzenie
Uderzyła ci woda sodowa do głowy
Uderzyła ci woda sodowa do głowy
Re: Wybarwianie roślin
Ciekawe jak inne rośliny zareagowały by na sod i chlorki i jakie setezenie było by optymalne w akwarium
Re: Wybarwianie roślin
Jakie rosliny z fotosynteza C4 ? ludziska nie dorabiajcie sobie tezy do czegos czego nie ma.
Tak juz jak kiedys pisalem i Michal wie,zresta juz sam wspomnial istnieje wykaz roslin z fotosynteza C4 i w tym wykazie widniej Michala popularna roslina,a mianowicie moczarka(Egeria) i tyle!!! nie ma wiecej roslin popularnych akwarystycznych,
Rozmawialem ostatnio u siebie z botanikiem ktory rowniez potwierdzil tozsamy spis roslin.
Tak jak pisalem nie raz na anglojezycznych stronach jest duzo roznych linkow do naukowych artykulow do ktorych przez lata korzystalem.
Dlatego powtarzam nie dorabiajmy sobie jeakiejs tezy na wlasny uzytek pod wlasna teze bez nauki. w tym aspekcie
O Na? prosze bardzo
Plant scientists usually classify plant mineral nutrients based on the concept of "essentiality" defined by Arnon and Stout as those elements necessary to complete the life cycle of a plant. Certain other elements such as Na have a ubiquitous presence in soils and waters and are widely taken up and utilized by plants, but are not considered as plant nutrients because they do not meet the strict definition of "essentiality." Sodium has a very specific function in the concentration of carbon dioxide in a limited number of C 4 plants and thus is essential to these plants, but this in itself is insufficient to generalize that Na is essential for higher plants. The unique set of roles that Na can play in plant metabolism suggests that the basic concept of what comprises a plant nutrient should be reexamined. We contend that the class of plant mineral nutrients should be comprised not only of those elements necessary for completing the life cycle, but also those elements which promote maximal biomass yield and/or which reduce the requirement (critical level) of an essential element. We suggest that nutrients functioning in this latter manner should be termed "functional nutrients." Thus plant mineral nutrients would be comprised of two major groups, "essential nutrients" and "functional nutrients." We present an array of evidence and arguments to support the classification of Na as a "functional nutrient," including its requirement for maximal biomass growth for many plants and its demonstrated ability to replace K in a number of ways, such as being an osmoticium for cell enlargement and as an accompanying cation for long-distance transport. Although in this paper we have only attempted to make the case for Na being a "functional nutrient," other elements such as Si and Se may also confirm to the proposed category of "functional nutrients."
For example, various monovalent cations (Na + , Rb + , Cs + , and NH 4 + ) can substitute for K + in the basic functions of osmotic regulation (along with organic "compatible solute" molecules, such as proline, glycine betaine, polyamines, and sugar alcohols; Clarkson and Hanson, 1980;Leigh and Wyn Jones, 1984;Shabala, 2013;White, 2013;Luo et al., 2017), charge balancing (Danchin and Nikel, 2019), and enzyme activation (Nitsos and Evans, 1969;Nieves-Cordones et al., 2016a). In plants, the physicochemical similarities between K + and Na + (in terms of solubility, ionic character, and atomic and hydrated radii) allow Na + to substitute for K + to a significant extent when external [K + ] ([K + ] ext ) or K + transport is limiting (Gaymard et al., 1998;Spalding et al., 1999;Subbarao et al., 2003;Battie-Laclau et al., 2014;Kronzucker et al., 2013;Benito et al., 2014;Hedrich and Shabala, 2018; see Section 2.2). This is illustrated by the facultative ability of root cells to take up Na + when K + is low or unavailable (Horie et al., 2007;Schulze et al., 2012). ...
... Indeed, in the case of a plant growing on Na + -free media, [Na + ] cyt /[K + ] cyt would be essentially zero (and its inverse undefined). Nevertheless, in the context of Na + as a beneficial nutrient (Subbarao et al., 2003;Kronzucker et al., 2013), it might prove worthwhile to measure [Na + ] cyt /[K + ] cyt in species, both glycophytic and halophytic, when they demonstrate improved growth under non-toxic Na + regimes. An optimal [Na + ] cyt /[K + ] cyt could perhaps be discovered in such cases, which might assist in understanding the mechanism(s) by which low-to-moderate Na + provision can benefit growth (Subbarao et al., 2003;Gattward et al., 2012;Kronzucker et al., 2013). ...
... Nevertheless, in the context of Na + as a beneficial nutrient (Subbarao et al., 2003;Kronzucker et al., 2013), it might prove worthwhile to measure [Na + ] cyt /[K + ] cyt in species, both glycophytic and halophytic, when they demonstrate improved growth under non-toxic Na + regimes. An optimal [Na + ] cyt /[K + ] cyt could perhaps be discovered in such cases, which might assist in understanding the mechanism(s) by which low-to-moderate Na + provision can benefit growth (Subbarao et al., 2003;Gattward et al., 2012;Kronzucker et al., 2013). It should be noted that, on a tissue level, gross Na + concentrations and [Na + ]/[K + ] ratios are low in most plants, especially in leaves, except under saline (usually toxic) conditions, when Na + can be hyper-accumulated (Flam-Shepherd et al., 2018). ...
Therefore, soil tests for both Na and K could improve the decision support systems for K fertilizer management on low-K soils with varied Na supply. It is well documented that Na is beneficial to osmotic relations and mineral nutrition of some plant species, especially when K is present at suboptimal concentrations
High Na concentrations in the root zone can cause antagonistic effects on other cations like K, Ca, Mg and manganese (Mn) (Hasegawa et al. 2000), and significantly reduce the uptake of K by plants (Shahzad et al. 2012). However, when K is at suboptimal concentrations, low and moderate soil Na can be beneficial for plant growth
Because of its severe impact on agricultural production, research pertaining to understanding the effects of NaCl is mainly focused on analyzing the effects of high doses (more than 50 mM), while the potential beneficial effects of low doses are relatively less investigated. Sodium is a beneficial element for plants [6,7]. Na functions as an indispensable element for most C 4 species, as Na + is required for pyruvate import to the chloroplast, which supports the carbon fixation process in C 4 photosynthesis [7,8]. ...
... Sodium is a beneficial element for plants [6,7]. Na functions as an indispensable element for most C 4 species, as Na + is required for pyruvate import to the chloroplast, which supports the carbon fixation process in C 4 photosynthesis [7,8]. Nitrogen uptake and assimilation, as well as the activity of photosystem II, require Na in C 4 species [7]. ...
... Na functions as an indispensable element for most C 4 species, as Na + is required for pyruvate import to the chloroplast, which supports the carbon fixation process in C 4 photosynthesis [7,8]. Nitrogen uptake and assimilation, as well as the activity of photosystem II, require Na in C 4 species [7]. In addition to C 4 plants or halophytes, many C 3 plants have shown growth stimulation upon Na application [
n plants the principal electrolyte role, played by Na+ ions in animals, is taken over by potassium (K+) ions. Plants are very good at excluding sodium ions, despite their similarity to potassium ions.
Chloride is definitely an essential nutrient, but you need less than 2ppm.
There aren't any symptoms as such, because plants don't have a sodium requirement, it isn't essential, plants can grow without any. Some plants may grow better with some added sodium, for some it won't make any difference.\
On the other hand, it should not be too much of a problem using Sodium Bicarbonate to adjust alkalinity, but it depends on the plants. The range is vast. Florida lakes, for example have Na++ concentrations from about 10ppm inland to over 1000ppm in areas near the coast, so there is some adaptability. Other plants originating from areas where there is no coast or saline source may not be as adaptable. Generally it's best to avoid using water from a water softener which uses salt as these will have very high Sodium concentrations.
Przy okazji...
There are no negative effects of high potassium.
I routinely dose Potassium at values exceeding 100ppm K+ so it's a pointless waste of time worrying about K+ toxicity.
Here is a tank dosed at approximately 130ppm K+
If people want to worry then they always choose to worry about CO2 above everything else.
dalej prosze...fotosynteza
It is unnecessary to supersaturate the water with Oxygen in order to see pearling. Therefore, his claim that a single plant will not pearl just because it is sitting in a large volume of water is not valid. Pearling is a phenomenon which is based on the RATE of Oxygen evolution. Oxygen does not have a very good solubility in water, so if the evolution rate exceeds the dissolution rate the gas will form bubbles.
The rate at which Oxygen is produced is directly proportional to the rate of photosynthesis. In turn, the photosynthetic rate depends on many factors such as light intensity, CO2 availability, nutrient availability, temperature and overall plant health. Likewise, the solubility of Oxygen will depend on pressure and temperature.
So all these variables will combine to determine how quickly Oxygen is released into the water and whether the gas will dissolve immediately or not.
Also, the strategy of Oxygen release is not necessarily due to the toxicity of Oxygen. The evolution of Oxygen is really a side issue that occurs when the O.E.C (Oxygen Evolving Complex) in the chloroplast disassembles the water molecules into it's constituents Hydrogen protons (H+) and Oxygen. The O.E.C wants the H+ because these protons are used in the almost the same way as a battery. It stores the H+ protons and this potential to generate energy in the form of a phosphate enzyme called ATP (Adinosine TriPhosphate) ATP is a recyclable enzyme which all living things use to generate energy.
Surplus Oxygen produced by the chloroplasts O.E.C is sent to the roots to oxygenate both the roots themselves as well as the sediment. This is a symbiotic relationship between plants and microorganisms in the sediment. Nitrifying bacteria are aerobic and therefore, it is in the plants interest to supply Oxygen to these microbes to ensure their survival. There are other aerobic microbes that convert certain compounds to chemical forms that can then be used more effectively by the plant. The mere fact that microbes will use the Oxygen and will burn their food means that they will then release CO2 as a byproduct of their metabolism. This is CO2 that rooted plants can use.
Photosynthesis is broken into the
1) light dependent reactions:
... consist of Photosystem 2 (PS2) and Photosystem 1 (PS1) where PS2 sole responsibility is to absorb light and generate ATP (which requires a phosphate group) and NADPH - during this process, an electron is excited in chlorophyll (found in Thylakoid -which actually change orientation depending on light availability) and liberate it into the electron transport chain, moving it around to trigger several processes. That electron is replaced via < photolysis > which does not use CO2. As a result, pearling is merely an indicator that chlorophyll antennae are absorbing photons of light, exciting and donating chlorophyll electrons to the ETC, and photolysis is occuring to replace those electrons.
As a result, I think we will have the following:
Given a healthy leaf, high light and filter off = pearling regardless of anything
2) light independent reactions:
Calvin cycle where we generate sugar with CO2, ADP and NADP+ are regenerated.
Tak jak pisalem zawsze opieram sie o naukowe prace ,i przez mase lata korzystalem z zagranicznych portali gdzie sa ludzie z calego swiata,gdzie wypowidaja sie ludzie nauki czy tez sa linki do ciekawych artykulow jesli ktos chce bardziej w to wchodzic
Np.
I realized that my thought that *phosphate directly controls CO2 is actually true. When of photosynthesis occurs, it converts ADP to ATP:
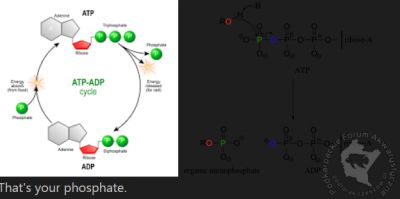
For our purposes, define low CO2 as concentration of CO2 < 30?, 28?, 25? 20? ... lets pick 20 for this exercise (go ahead and pick 30 since it is the proposed magical target - it makes no difference).
The photosynthetic rate of a plant is determined by both light intensity and available phosphate:
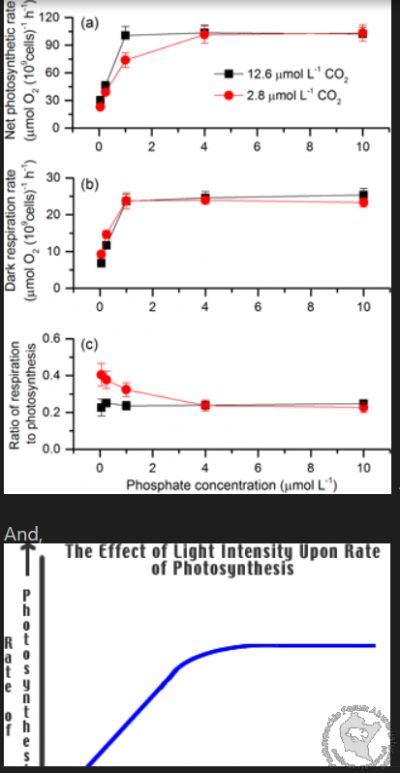
Tak juz jak kiedys pisalem i Michal wie,zresta juz sam wspomnial istnieje wykaz roslin z fotosynteza C4 i w tym wykazie widniej Michala popularna roslina,a mianowicie moczarka(Egeria) i tyle!!! nie ma wiecej roslin popularnych akwarystycznych,
Rozmawialem ostatnio u siebie z botanikiem ktory rowniez potwierdzil tozsamy spis roslin.
Tak jak pisalem nie raz na anglojezycznych stronach jest duzo roznych linkow do naukowych artykulow do ktorych przez lata korzystalem.
Dlatego powtarzam nie dorabiajmy sobie jeakiejs tezy na wlasny uzytek pod wlasna teze bez nauki. w tym aspekcie
O Na? prosze bardzo
Plant scientists usually classify plant mineral nutrients based on the concept of "essentiality" defined by Arnon and Stout as those elements necessary to complete the life cycle of a plant. Certain other elements such as Na have a ubiquitous presence in soils and waters and are widely taken up and utilized by plants, but are not considered as plant nutrients because they do not meet the strict definition of "essentiality." Sodium has a very specific function in the concentration of carbon dioxide in a limited number of C 4 plants and thus is essential to these plants, but this in itself is insufficient to generalize that Na is essential for higher plants. The unique set of roles that Na can play in plant metabolism suggests that the basic concept of what comprises a plant nutrient should be reexamined. We contend that the class of plant mineral nutrients should be comprised not only of those elements necessary for completing the life cycle, but also those elements which promote maximal biomass yield and/or which reduce the requirement (critical level) of an essential element. We suggest that nutrients functioning in this latter manner should be termed "functional nutrients." Thus plant mineral nutrients would be comprised of two major groups, "essential nutrients" and "functional nutrients." We present an array of evidence and arguments to support the classification of Na as a "functional nutrient," including its requirement for maximal biomass growth for many plants and its demonstrated ability to replace K in a number of ways, such as being an osmoticium for cell enlargement and as an accompanying cation for long-distance transport. Although in this paper we have only attempted to make the case for Na being a "functional nutrient," other elements such as Si and Se may also confirm to the proposed category of "functional nutrients."
For example, various monovalent cations (Na + , Rb + , Cs + , and NH 4 + ) can substitute for K + in the basic functions of osmotic regulation (along with organic "compatible solute" molecules, such as proline, glycine betaine, polyamines, and sugar alcohols; Clarkson and Hanson, 1980;Leigh and Wyn Jones, 1984;Shabala, 2013;White, 2013;Luo et al., 2017), charge balancing (Danchin and Nikel, 2019), and enzyme activation (Nitsos and Evans, 1969;Nieves-Cordones et al., 2016a). In plants, the physicochemical similarities between K + and Na + (in terms of solubility, ionic character, and atomic and hydrated radii) allow Na + to substitute for K + to a significant extent when external [K + ] ([K + ] ext ) or K + transport is limiting (Gaymard et al., 1998;Spalding et al., 1999;Subbarao et al., 2003;Battie-Laclau et al., 2014;Kronzucker et al., 2013;Benito et al., 2014;Hedrich and Shabala, 2018; see Section 2.2). This is illustrated by the facultative ability of root cells to take up Na + when K + is low or unavailable (Horie et al., 2007;Schulze et al., 2012). ...
... Indeed, in the case of a plant growing on Na + -free media, [Na + ] cyt /[K + ] cyt would be essentially zero (and its inverse undefined). Nevertheless, in the context of Na + as a beneficial nutrient (Subbarao et al., 2003;Kronzucker et al., 2013), it might prove worthwhile to measure [Na + ] cyt /[K + ] cyt in species, both glycophytic and halophytic, when they demonstrate improved growth under non-toxic Na + regimes. An optimal [Na + ] cyt /[K + ] cyt could perhaps be discovered in such cases, which might assist in understanding the mechanism(s) by which low-to-moderate Na + provision can benefit growth (Subbarao et al., 2003;Gattward et al., 2012;Kronzucker et al., 2013). ...
... Nevertheless, in the context of Na + as a beneficial nutrient (Subbarao et al., 2003;Kronzucker et al., 2013), it might prove worthwhile to measure [Na + ] cyt /[K + ] cyt in species, both glycophytic and halophytic, when they demonstrate improved growth under non-toxic Na + regimes. An optimal [Na + ] cyt /[K + ] cyt could perhaps be discovered in such cases, which might assist in understanding the mechanism(s) by which low-to-moderate Na + provision can benefit growth (Subbarao et al., 2003;Gattward et al., 2012;Kronzucker et al., 2013). It should be noted that, on a tissue level, gross Na + concentrations and [Na + ]/[K + ] ratios are low in most plants, especially in leaves, except under saline (usually toxic) conditions, when Na + can be hyper-accumulated (Flam-Shepherd et al., 2018). ...
Therefore, soil tests for both Na and K could improve the decision support systems for K fertilizer management on low-K soils with varied Na supply. It is well documented that Na is beneficial to osmotic relations and mineral nutrition of some plant species, especially when K is present at suboptimal concentrations
High Na concentrations in the root zone can cause antagonistic effects on other cations like K, Ca, Mg and manganese (Mn) (Hasegawa et al. 2000), and significantly reduce the uptake of K by plants (Shahzad et al. 2012). However, when K is at suboptimal concentrations, low and moderate soil Na can be beneficial for plant growth
Because of its severe impact on agricultural production, research pertaining to understanding the effects of NaCl is mainly focused on analyzing the effects of high doses (more than 50 mM), while the potential beneficial effects of low doses are relatively less investigated. Sodium is a beneficial element for plants [6,7]. Na functions as an indispensable element for most C 4 species, as Na + is required for pyruvate import to the chloroplast, which supports the carbon fixation process in C 4 photosynthesis [7,8]. ...
... Sodium is a beneficial element for plants [6,7]. Na functions as an indispensable element for most C 4 species, as Na + is required for pyruvate import to the chloroplast, which supports the carbon fixation process in C 4 photosynthesis [7,8]. Nitrogen uptake and assimilation, as well as the activity of photosystem II, require Na in C 4 species [7]. ...
... Na functions as an indispensable element for most C 4 species, as Na + is required for pyruvate import to the chloroplast, which supports the carbon fixation process in C 4 photosynthesis [7,8]. Nitrogen uptake and assimilation, as well as the activity of photosystem II, require Na in C 4 species [7]. In addition to C 4 plants or halophytes, many C 3 plants have shown growth stimulation upon Na application [
n plants the principal electrolyte role, played by Na+ ions in animals, is taken over by potassium (K+) ions. Plants are very good at excluding sodium ions, despite their similarity to potassium ions.
Chloride is definitely an essential nutrient, but you need less than 2ppm.
There aren't any symptoms as such, because plants don't have a sodium requirement, it isn't essential, plants can grow without any. Some plants may grow better with some added sodium, for some it won't make any difference.\
On the other hand, it should not be too much of a problem using Sodium Bicarbonate to adjust alkalinity, but it depends on the plants. The range is vast. Florida lakes, for example have Na++ concentrations from about 10ppm inland to over 1000ppm in areas near the coast, so there is some adaptability. Other plants originating from areas where there is no coast or saline source may not be as adaptable. Generally it's best to avoid using water from a water softener which uses salt as these will have very high Sodium concentrations.
Przy okazji...
There are no negative effects of high potassium.
I routinely dose Potassium at values exceeding 100ppm K+ so it's a pointless waste of time worrying about K+ toxicity.
Here is a tank dosed at approximately 130ppm K+
If people want to worry then they always choose to worry about CO2 above everything else.
dalej prosze...fotosynteza
It is unnecessary to supersaturate the water with Oxygen in order to see pearling. Therefore, his claim that a single plant will not pearl just because it is sitting in a large volume of water is not valid. Pearling is a phenomenon which is based on the RATE of Oxygen evolution. Oxygen does not have a very good solubility in water, so if the evolution rate exceeds the dissolution rate the gas will form bubbles.
The rate at which Oxygen is produced is directly proportional to the rate of photosynthesis. In turn, the photosynthetic rate depends on many factors such as light intensity, CO2 availability, nutrient availability, temperature and overall plant health. Likewise, the solubility of Oxygen will depend on pressure and temperature.
So all these variables will combine to determine how quickly Oxygen is released into the water and whether the gas will dissolve immediately or not.
Also, the strategy of Oxygen release is not necessarily due to the toxicity of Oxygen. The evolution of Oxygen is really a side issue that occurs when the O.E.C (Oxygen Evolving Complex) in the chloroplast disassembles the water molecules into it's constituents Hydrogen protons (H+) and Oxygen. The O.E.C wants the H+ because these protons are used in the almost the same way as a battery. It stores the H+ protons and this potential to generate energy in the form of a phosphate enzyme called ATP (Adinosine TriPhosphate) ATP is a recyclable enzyme which all living things use to generate energy.
Surplus Oxygen produced by the chloroplasts O.E.C is sent to the roots to oxygenate both the roots themselves as well as the sediment. This is a symbiotic relationship between plants and microorganisms in the sediment. Nitrifying bacteria are aerobic and therefore, it is in the plants interest to supply Oxygen to these microbes to ensure their survival. There are other aerobic microbes that convert certain compounds to chemical forms that can then be used more effectively by the plant. The mere fact that microbes will use the Oxygen and will burn their food means that they will then release CO2 as a byproduct of their metabolism. This is CO2 that rooted plants can use.
Photosynthesis is broken into the
1) light dependent reactions:
... consist of Photosystem 2 (PS2) and Photosystem 1 (PS1) where PS2 sole responsibility is to absorb light and generate ATP (which requires a phosphate group) and NADPH - during this process, an electron is excited in chlorophyll (found in Thylakoid -which actually change orientation depending on light availability) and liberate it into the electron transport chain, moving it around to trigger several processes. That electron is replaced via < photolysis > which does not use CO2. As a result, pearling is merely an indicator that chlorophyll antennae are absorbing photons of light, exciting and donating chlorophyll electrons to the ETC, and photolysis is occuring to replace those electrons.
As a result, I think we will have the following:
Given a healthy leaf, high light and filter off = pearling regardless of anything
2) light independent reactions:
Calvin cycle where we generate sugar with CO2, ADP and NADP+ are regenerated.
Tak jak pisalem zawsze opieram sie o naukowe prace ,i przez mase lata korzystalem z zagranicznych portali gdzie sa ludzie z calego swiata,gdzie wypowidaja sie ludzie nauki czy tez sa linki do ciekawych artykulow jesli ktos chce bardziej w to wchodzic
Np.
I realized that my thought that *phosphate directly controls CO2 is actually true. When of photosynthesis occurs, it converts ADP to ATP:

For our purposes, define low CO2 as concentration of CO2 < 30?, 28?, 25? 20? ... lets pick 20 for this exercise (go ahead and pick 30 since it is the proposed magical target - it makes no difference).
The photosynthetic rate of a plant is determined by both light intensity and available phosphate:

Re: Wybarwianie roślin
Zaciekawiło mnie to z tym potasem, tak wysoko potas a zawsze była mowa że potas to bloker np wapnia sam Rataj o tym mówi, że potas nie może być powyżej wapnia bo blokuje, wapń, magnez, sód i bor.
Re: Wybarwianie roślin
Wiesz lubie Rataja bo tak jak on czy Drak sa profesorami/doktorami z zakresu roslin i wiem ze Rataj akurat ma taka teze,niechce mi sie szukac ale np. chcialbym uslyszec uzasadnienie dlaczego...,skoro np. co mowi nam sucha masa rosliny ile jest K a ile jest Ca czy Mg ,wiec nawet w suchej masie K jest wyzej dla zdrowego wzrostu/zapotrzebowania .
Jak juz kiedys podawalem osoba ktora ,przez jeden rok testowala jeden skladnik i w kolejnym nastepny
tylko dla jednego skladnika poswiecala wszystkie zaleznosci itd.
prowadzila zbiornik na
Ca -4.8
Mg- 0.8
k -16
tam niebylo zadnych problemow.
Jak juz kiedys podawalem osoba ktora ,przez jeden rok testowala jeden skladnik i w kolejnym nastepny
tylko dla jednego skladnika poswiecala wszystkie zaleznosci itd.
prowadzila zbiornik na
Ca -4.8
Mg- 0.8
k -16
tam niebylo zadnych problemow.
Re: Wybarwianie roślin
Wszystkie Marek, wszystkie.
Uczepiłeś się sodu a tutaj problem sprowadza się do magnezu.
Ja nie będę wklejał linków do mega opracowań których i tak miałbym problem z jego obroną. Sugeruję jedynie poznanie analizy budowy liścia korzystającego z fotosyntezy C3 a C4.
Byłem pewny, że wklejałem linka z którego ja korzystałem ale znalazłem to
upload/u10_1425983082.pdf
8 strona pdf. Jedno zdanie na temat C4. Nad tym boleje.
Zanim napiszę elaborat napisz jak podchodzisz do roli magnezu w nawożeniu roślin wodnych.
Prot, dla mnie pełzacz jest wskaźnikowy jeżeli chodzi o molibden czyli nadmiar siarczanów.
Pozdrawiam
Michał
Michał
- duch amazonski
- Posty: 1932
- Rejestracja: 03 wrz 2014, 19:44
- Imię: Darek
- Lokalizacja: LUBLIN
- Płeć:
- Wiek: 49
Re: Wybarwianie roślin
Ciekawy artykuł.
https://www.2hraquarist.com/blogs/fresh ... red-plants
https://www.2hraquarist.com/blogs/fresh ... red-plants
Prawdziwy mężczyzna płacze tylko wtedy, gdy szampon szczypie w oczka.